SITC 2025 | Single Intravenous Infusion Leads to Complete Clearance of High-Grade Glioblastoma in 4 Weeks
The 40th Annual Meeting of the Society for Immunotherapy of Cancer (SITC) will officially kick off in Maryland, USA, from November 7 to 9, 2025. As one of the world's largest and most prestigious international conferences focused on cancer immunotherapy, the SITC Annual Meeting is dedicated to advancing the science and application of cancer immunotherapy, discovering breakthroughs, and improving the prognosis for all cancer patients.
Juncell Therapeutics' latest clinical breakthrough with GC101 has also been included. A patient with grade IV glioblastoma achieved complete tumor clearance just 4 weeks after a single infusion of GC101 TILs, and has maintained tumor-free survival for more than 20 months to date—bringing new hope for the treatment of such advanced malignant tumors. This latest breakthrough will be shared with global industry experts in the form of a poster presentation and online session.
Poster Details
As one of the most common primary intracranial tumors, glioblastoma is regarded as a tough nut to crack in the field of oncology due to its high malignancy and strong heterogeneity. Traditional treatment combining surgery, radiotherapy, and chemotherapy not only struggles to eradicate lesions but also causes severe side effects. The median survival time of patients is 6-10 months, with a 5-year survival rate of less than 10%. More challenging still, the blood-brain barrier (BBB) acts as a natural barrier, blocking most drugs and severely compromising therapeutic efficacy.
Leveraging Juncell Therapeutics' independently developed DeepTIL™ technology platform, its innovative time-segmented process has achieved key technological breakthroughs. Tumor tissue is obtained from patients during initial surgery to pre-manufacture and preserve TIL seed cells. Upon recurrence, the cells are rapidly resuscitated, cultured, and infused—shortening waiting time while retaining highly active killer cells.
Conventional IL-2-dependent TIL therapy for glioblastoma has certain limitations: on one hand, it is quite challenging to cultivate clinical-grade TIL cells from cold tumors; on the other hand, IL-2 injection may increase the risk of severe adverse reactions such as cerebral edema and cerebral hemorrhage.
Juncell's innovative GC101 TIL therapy eliminates the need for high intensity lymphodepletion or the use of IL-2 injections. It can independently cross the blood-brain barrier after intravenous infusion to precisely kill tumors, offering the advantages of rapid onset, long-lasting efficacy, and mild side effects. This breakthrough in glioblastoma treatment represents another significant advancement of GC101 following its success in pancreatic cancer, melanoma, lung cancer, gynecologic cancers, and other cancer types, which is expected to completely reshape the treatment landscape for glioblastoma.
From Despair to Hope
At the critical juncture of his life, Mr. W first underwent a high-risk brain tumor resection. Following the surgery, he received temozolomide chemotherapy and radiotherapy, but his condition was not controlled. Subsequently, he tried two CAR-T therapies targeting different antigens, completing more than 10 intratumoral injections in total, yet still failed to continuously halt tumor progression.
Patient's Treatment History
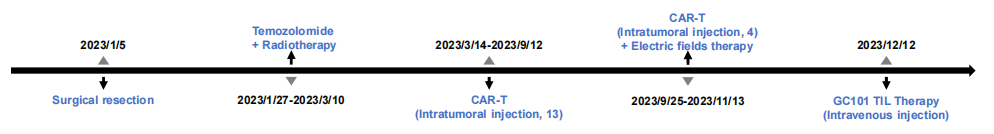
At the end of 2023, as Mr. W and his family fell into despair, a turn of fate quietly arrived. Mr. W successfully enrolled in the GC101 clinical trial and received TIL cell reinfusion therapy (the cells were derived from tumor tissue obtained and cultured during his surgery one year ago). Only 4 weeks after infusion, imaging results showed complete clearance of his brain tumor, with the therapeutic effect evaluated as Complete Response (CR). No recurrence has been observed since then, and he has maintained a tumor-free survival for more than 20 months. Today, Mr. W has long been free from hospital beds, reborn to return to a normal life.
Patient's MRI Image Changes
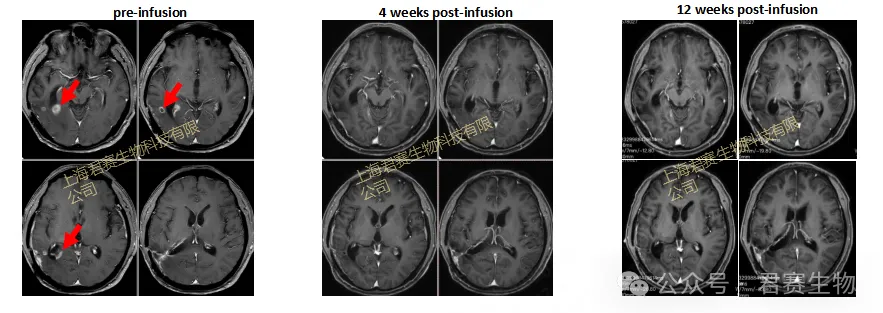
To date, this case has not only set a new survival record for recurrent glioblastoma but also revalidated the durable efficacy of TIL therapy with compelling clinical data. Meanwhile, it has provided critical clinical evidence for the treatment strategy of tumor tissue collection at an early stage and TIL reinfusion upon recurrence, bringing the hope of life to countless solid tumor patients in desperate situations.
