Robust
Reproducible performance across over 30 types of solid tumors. Culture success rate >95%
Competent
The average number of cells is approximately 20 billion, IL-2 administration free
Affordable
Substantial reduction in manufacturing costs and overall treatment costs
Accessible
Patients treated in less demanding wards, and the time to reach discharge criteria was shortened

Technological Platforms
-
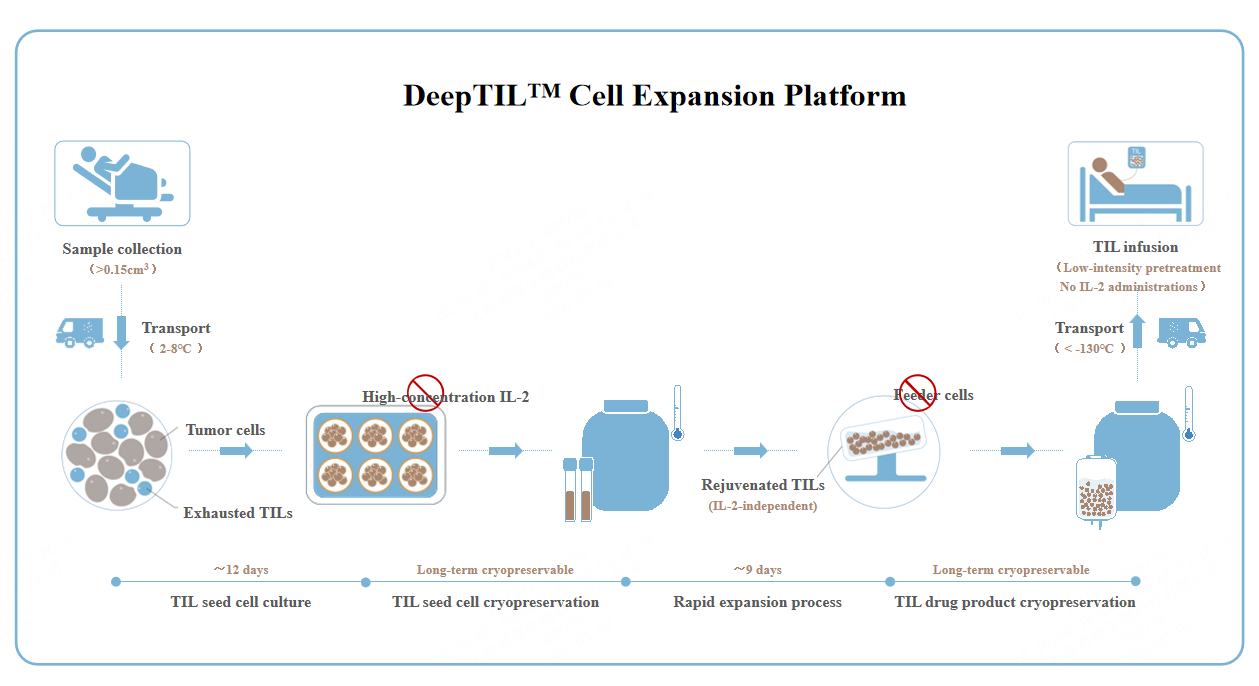
DeepTILTMCell Expansion Platform
-
Highly Efficient TIL Enrichment for Pan-solid Tumors
Clinical-grade TILs can be obtained with a high successful rate.
-
Simplified Process Without Feeder Cells
TIL cell culture does not rely on healthy human PBMC as feeder cells, resulting in a simpler process and lower cost.
-
No High-intensity Lymphodepletion Chemotherapy or Use of IL-2
Free from high-intensity lymphodepletion chemotherapy and IL-2 administration, treatment can be delivered in a general ward, with improved safety.
-
-
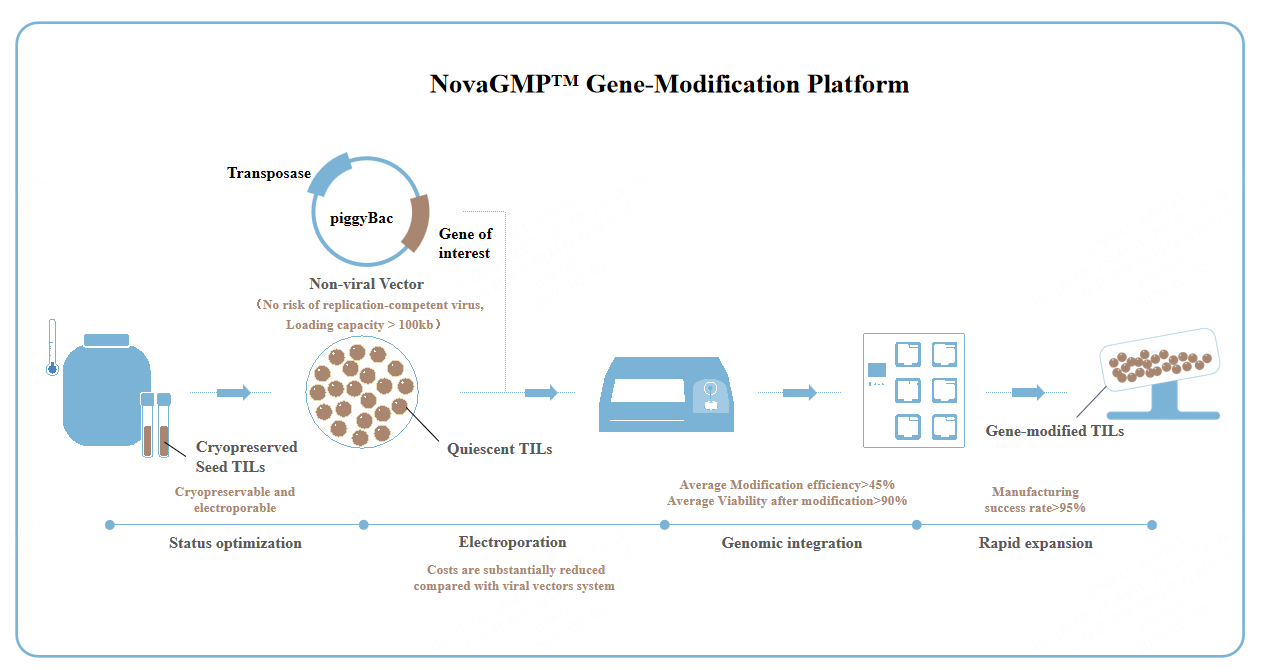
NovaGMPTM Gene Modification Platform
-
Safer
Non-viral gene modification. No risk of wild-type virus mutations.
-
More Affordable
The cost of non-viral vector-based gene modification is substantially reduced compared with that of viral vectors system.
-
More Efficient
Average gene modification efficiency >45%, Average viability after modification >90%
-
-
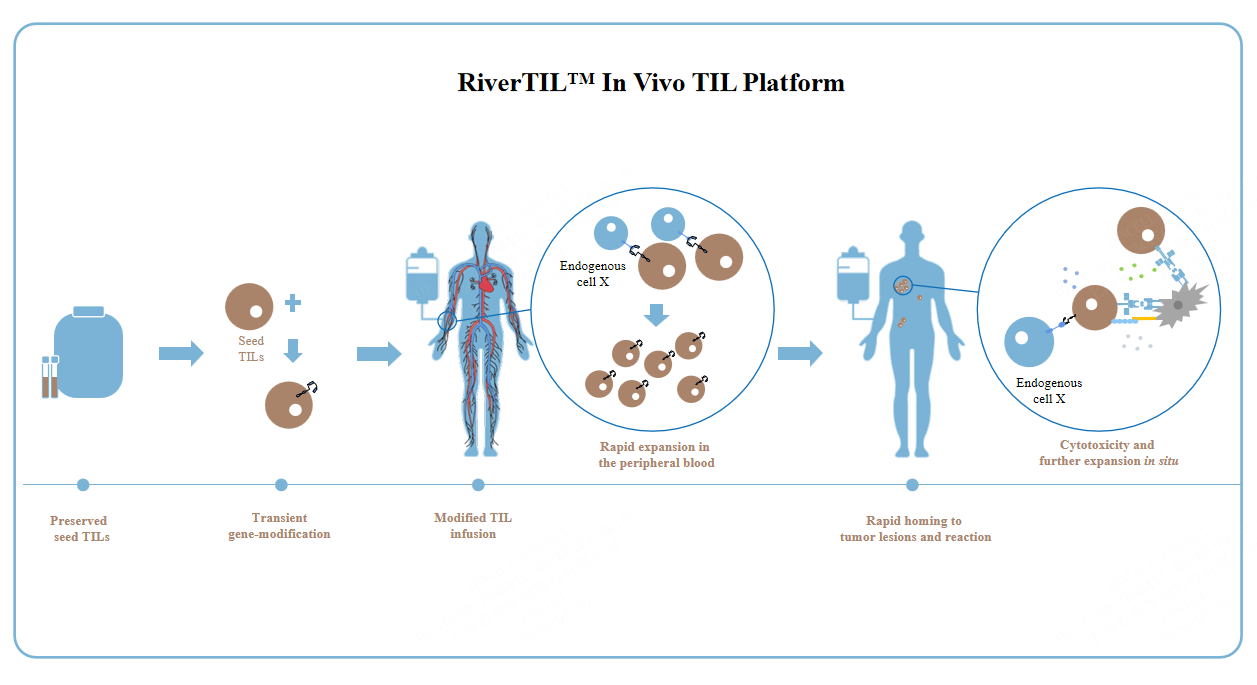
RiverTIL™ In Vivo TIL Platform
-
Time-segmented Process
Pre-manufacture and cryopreserve TIL seed cells from a patient's resected tumor tissue for future on-demand use.
-
Transient Non-viral Gene Modification
Rapid in vivo expansion and reaction after seed TIL infusion.
-
Improved Accessibility
Significantly reducing manufacturing costs and production time.
-


